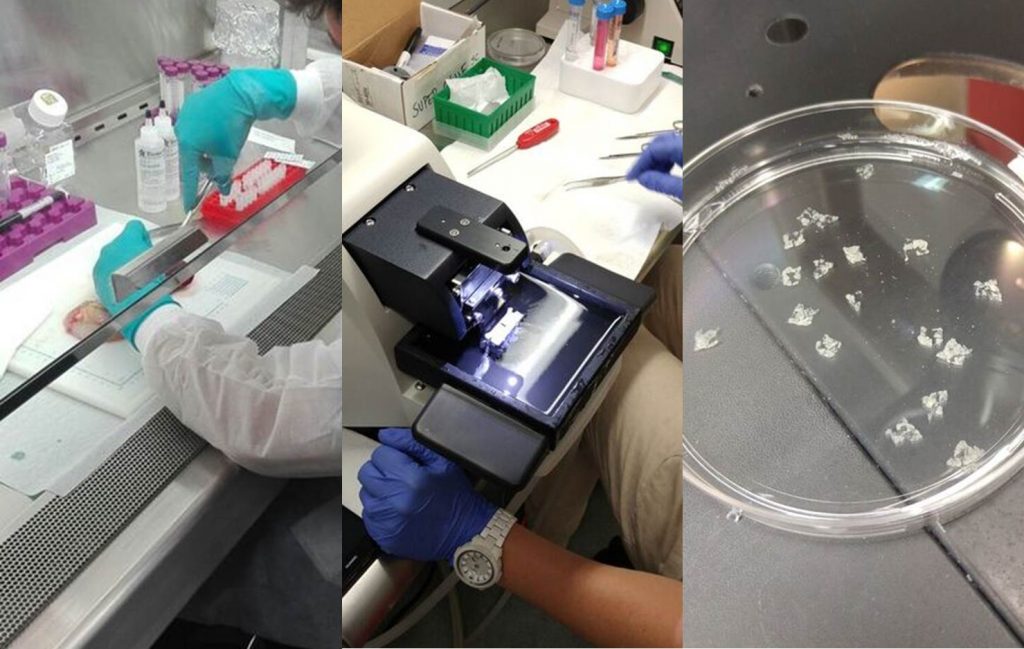
Dr. Stephan Speier has recently established a novel technique for generating pancreas tissue slices, which are suitable for conducting functional assessments of beta cells without the need for and the downsides of islet isolation (Marciniak et al, Nat Protoc, 2014). This technique has been developed in rodent pancreas, and Dr. Speier had conducted limited experimentation with human tissue from pancreatic resections. At nPOD we felt that this approach would be highly valuable to obtain a live readout of several aspects of beta cell function from the pancreas of nPOD organ donors. We decided to investigate whether indeed it was feasible to apply this novel methodology to the study of organ donors, which unlike tissue from resections are perfused and shipped in preservative solutions, and have several hours of cold ischemia time.
To this end, and with support from the Helmsley Charitable Trust George Eisenbarth nPOD Award for Team Science, Dr. Speier and Dr. Julia Stertmann traveled to the Diabetes Research Institute in Miami (home of the nPOD Co-Executive Director, Dr. Alberto Pugliese), where the required instrument (vibratome) to generate tissue slices was available (it was purchased for this purpose with support from Dr. Camillo Ricordi and the Diabetes Research Institute Foundation). The whole nPOD team from Gainesville was engaged in this activity: this required a high level of coordination to procure high quality pancreata, have them shipped to Miami, processed there under nPOD SOPs and then shipped back (fixed or frozen) to the nPOD OPPC. nPOD’s own Stephen Selman spent a month in Miami, overseeing the processing of the organs. Together with Dr. Sirlene Cechin from the Pugliese lab, they learned the technique for producing pancreas slices. After slices were generated, they were perfused, cultured, assessed with functional tests and imaged, for which we also relied on the expertise of nPOD Investigator Dr. Alejandro Caicedo (Department of Medicine, Division of Endocrinology, University of Miami).
Several organs were received and processed during this period, and many lessons learned. Importantly, the team was able to generate pancreas slices from organ donors. Such slices remained viable for at least 48 hours (plain tissue fragments did not), and after overnight shipment. Insulin secretion was stimulated and measured from the pancreas slices and images were acquired.
Having shown the basic feasibility of this approach, Drs. Speier and Pugliese are developing plans for a collaborative working group that will further develop this approach, including the extension of viability over longer periods of culture. This would allow employing this approach for addressing important research questions that require a longer incubation time to observe effects (for example, viral infections). We also envision this approach being applied routinely by nPOD to have a baseline functional assessment that would be relevant to nPOD researchers as they may correlate their findings with the assessed islet function of each donor. This should be much more informative than serum C-peptide levels. Once the system is fully established at the nPOD OPPC, we hope it will be possible for nPOD investigators to receive living pancreas slices that they can use in their own functional studies. Stay tuned for updates, we will share more information about this in the coming months. If you have questions or suggestions, or are interested, please email Joseph Pobee, pobee@ufl.edu, your nPOD Investigator Coordinator.
 Stephan Speier, PhD
Stephan Speier, PhD
Group Leader, Islet Cell Physiology
Paul Langerhans Institute Dresden (PLID) of Helmholtz Center Munich at
the University Clinic Carl Gustav Carus of Technische Universität Dresden,
Helmholtz Zentrum München, Neuherberg
German Center for Diabetes Research (DZD)
DFG-Center for Regenerative Therapies Dresden (CRTD), Faculty of
Medicine, Technische Universität Dresden, Germany